Abstract
Introduction: Prognosis of patients (pts) with myelodysplastic syndrome (MDS), particularly the group with lower-risk disease (LR-MDS) is very heterogeneous. In the last decade, different studies have reported the role of recurrent somatic mutations involved in multiple cellular pathways and incorporated them into clinical practice and current prognostic models, including all risk categories. Recently, the incorporation of genomic data to clinical parameters defined the new Molecular International Prognostic Scoring System (IPSS-M).
Materials and Methods: A retrospective study evaluating the impact of molecular profile in a series of 181 pts with LR-MDS and non-proliferative chronic myelomonocytic leukemia (CMML) focused on survival estimates and the probability of leukemic progression. Genomic DNA was isolated from bone marrow mononuclear cells at diagnosis using DNA kit from Qiagen according to the manufacturer´s instructions. A PCR-based targeted was performed sequencing of 40 genes involved in myeloid neoplasms (Oncomine Myeloid Research panel, including hotspots of ABL, BRAF, CBL, CSF3R, DNMT3A, FLT3, GATA2, HRAS, IDH1, IDH2, JAK2, KIT, KRAS, MPL, MYD88, NPM1, NRAS, PTPN11, SETBP1, SF3B1, SRSF2, U2AF1, WT1 and entire coding regions of ASXL1, BCOR, CALR, CEBPA, ETEV6, EZH2, IKZF1, NF1, PHF6, PRPF8, RB1, RUNX1, SH2B3, STAG2, TET2, TP53, ZRSR2). Sequencing was performed using the Ion Torrent S5 sequencing platform from Life Technologies. Survival (OS) was analyzed from diagnosis until death as a result of any cause or the last follow-up and calculated by the Kaplan-Meier method and log rank test. A Cox proportional hazard regression model was performed to analyze OS as endpoint. Analysis was performed by the IBMÒ SPSSÒ version 26.0.0. The probability of leukemic progression was analyzed using the Gray test in the framework of competing risk and considering death not attributable to AML as a competing event. This study was conducted at Hospital Universitario Virgen del Rocío, approved by the local ethics committee and was performed in accordance with the Declaration of Helsinki.
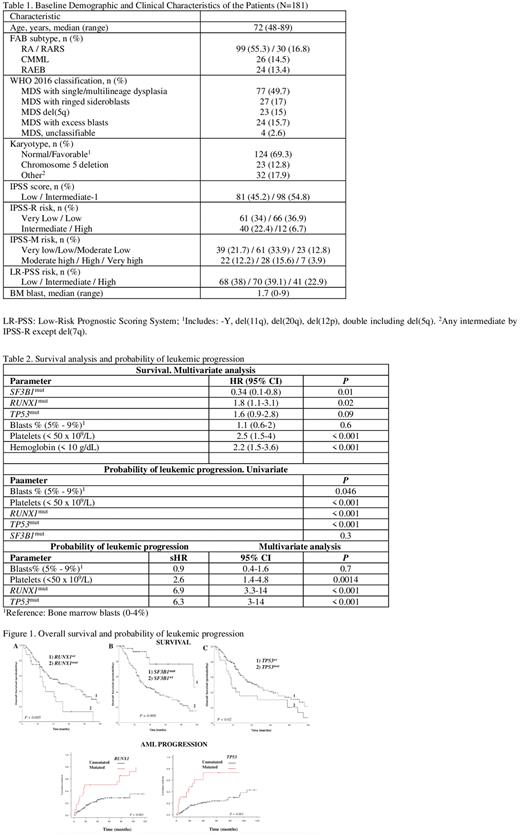
Results: In total, 392 mutations were detected in the 181 pts analyzed with a median of 2 mutations per sample (range: 0-8). Sixty-seven pts (37%) harbored ³3 mutations. Table shows baseline characteristics of pts. Most frequent mutated genes were TET2, SF3B1, RUNX1, ASXL1, TP53, and SRSF2 (all of them present in >10% of pts). Epigenetic regulation was the most recurrently mutated pathway, followed by mutations in splicing factors. After a median follow-up of 47 months (95% CI, 37-56 months), 93 out of 191 pts had died (48%). Median OS for the whole cohort was 41 months (95% CI, 28-53 months). TP53 mutations were detected in 22 pts, with equal distribution of mono-allelic or multi-hit TP53 state. As no significant differences in median OS was observed between groups [23 months (95% CI, 12-53 months) in mono-allelic TP53 state vs. 17 months (95% CI, 11-22 months) in multi-hit cases ( P = 0.1)], TP53 mutation was analyzed as a single parameter. Mutations in SF3B1 were detected in 34 pts and the majority of them (31 out of 34; 91%) were classified as SF3B1a according to the recent segregation of SF3B1-mutated cases by the IPSS-M. In univariate analysis, RUNX1 or TP53mutations correlated with lower median OS (Table 2). In contrast, SF3B1 mutation was associated with prolonged median OS [95 months (95% IC, 32-157) vs. 33 months (95% CI, 19-46) in unmutated pts (P < 0.01)]. In a multivariate Cox regression model, RUNX1 mutations independently associated with shorter OS, while SF3B1 mutation retained its favorable impact on outcome (HR: 0.24, 95% CI, 0.1-0.5; P = 0.001). At last follow-up, 49 patients (25%) progressed to AML. Median time to progression was 16.2 months (range: 2-105 months). Subdistribution hazard ratios (sHRs) for leukemic progression were estimated to identify predictive covariates for progression. TP53 mutation (sHR, 6.3; 95% CI, 3-14); P < 0.001) and RUNX1 mutation (sHR, 6.9; 95% CI, 3.3-14; P < 0.001) were predictive for the probability of leukemic progression (Table 2).
Conclusion: Incorporation of molecular testing in LR-MDS identified a subset of pts with expected poorer outcome, either due to lower survival or probability of leukemic progression. Results herein reported may help prognosis stratification in this set of pts with low-risk disease and support the recommendation of molecular testing in pts with MDS.
Disclosures
Rodriguez Arbolí:Jazz Pharmaceuticals: Consultancy; MSD: Consultancy; Pfizer: Honoraria, Other: travel/conference grants; Eurocept: Consultancy. Perez-Simon:Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, and Expenses; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, and Expenses; GILEAD: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, and Expenses; JAZZ: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, and Expenses; ALEXION: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, and Expenses; ABBVIE: Research Funding; PFIZER: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal